Regulatory Toxicology - Permitted Daily Exposure (PDE)
Regulatory Toxicology
Ensuring Product Safety & Compliance
Comprehensive toxicology solutions for regulatory approval, product safety, and global market access across diverse industries.

Risk Assessment Excellence
Expert toxicological evaluations to identify, assess, and mitigate product safety risks for regulatory success.

Global Regulatory Support
Guiding products through complex regulations with tailored compliance strategies and documentation.

Lifecycle Safety Management
Ongoing toxicology services to support product development, approval, and global post-market access.


Edit Content
We help ensure patient safety by evaluating extractables, leachables, and excipients for toxicological risk
Evaluation of chemical substances migrating from packaging or device components and assessment of
excipients used in formulations for toxicological safety. Required in pharmaceuticals and medical
devices under USP 1663/1664, ISO 10993, and excipient safety assessments in cosmetic and
nutraceutical formulations

Edit Content
We help you meet global limits for toxic elements and residual solvents in final products.
Risk-based analysis of trace metals (ICH Q3D) and organic solvents (ICH Q3C) remaining in the final
product. Mandatory for pharmaceuticals and APIs; essential for nutraceuticals, cosmetics, and
medical devices to ensure product quality and safety

Edit Content
We protect manufacturing personnel through scientifically derived occupational exposure limits.
Determination of airborne concentration limits to safeguard workers handling active substances or
chemicals. Required for pharmaceutical and API production, and increasingly adopted for chemicals
used in device and cosmetic manufacturing.

Edit Content
We help to establish safe exposure limits for humans across all product categories.
Scientific determination of safe daily exposure levels for humans based on toxicology data.
Applicable across all regulated industries to justify ingredient or impurity safety.

Edit Content
We ensure global compliance through accurate product classification and labeling documentation.
Preparation of product classification (e.g., GHS, CLP) and EHS data for regulatory compliance and
hazard communication. Required for shipping, workplace safety, and registration in all sectors.
Edit Content
We validate cleaning procedures and assess cross-contamination risk in manufacturing facilities.
Use of toxicological limits (e.g., PDE) to evaluate cleaning effectiveness and prevent crosscontamination between products. Mandatory in GMP environments, especially for pharmaceutical and
API manufacturing.

Edit Content
We help you to quantify consumer risk from multiple exposure sources using MoS calculations
Calculation of safety margins by comparing systemic exposure to toxicological thresholds; includes
multiple ingredient or route exposures. Required in cosmetic product safety reports, nutraceutical
evaluations, and impurity profiling in pharmaceuticals.

Edit Content
We provide compliant SDS authoring and regulatory reviews for global supply chains.
Authoring and reviewing SDSs in line with global chemical regulations (GHS, REACH, OSHA).
Permissible requirement for raw materials, ingredients, and finished products across all industries.

Edit Content
We model realistic human exposure based on the intended route of administration
Modeling and quantifying human exposure via ingestion, skin contact, or inhalation based on intended
use. Supports risk assessments for medical devices, cosmetics, drugs, and food supplements.

Edit Content
We establish the toxicological identity of substances for accurate hazard communication.
Characterization of toxicological properties (e.g., genotoxicity, sensitization, irritation) to support
hazard classification. Integral to SDS authoring, labeling, and regulatory submissions.

Edit Content
We help meet environmental safety requirements through robust ERA documentation.
Evaluation of environmental fate, persistence, and toxicity to aquatic and terrestrial species. Required
for pharmaceuticals (EMA), cosmetics, agrochemicals, and industrial chemicals.

Edit Content
We determine health-based exposure limits to support cleaning validation and containment strategies
Scientific derivation of Permitted Daily Exposure (PDE), Acceptable Daily Exposure (ADE), and
Health-Based Exposure Limits (HBEL). Required for GMP cleaning validation and containment
policy in pharma and medical devices.

Edit Content
We calculate toxicologically justified F-values for cleaning validation programs.
Toxicologically derived visual residue limit (F-value) for use in cleaning validation and visual
inspection protocols. Supports GMP compliance in pharmaceutical manufacturing.

Edit Content
Molecular Characterization
We identify and define molecular hazards to support early-stage product development. In silico
evaluation of molecular structures for hazard identification and classification. Critical for preclinical
development and substance registration

Edit Content
Cosmetics & Personal Care Regulatory Submissions
We ensure global market access through compliant cosmetic product submissions. Development of
safety assessments, Product Information Files (PIF), and CPNP documents. Mandatory under EU
Regulation 1223/2009 and global equivalents.

Edit Content
We support ingredient approvals through scientific and regulatory justification.
Toxicological and exposure-based safety assessments for novel food ingredients and dietary
supplements. Required by EFSA, FDA, FSSAI, and other food safety authorities.

Edit Content
We help achieve ISO 10993 compliance for biocompatibility and toxicological safety of medical devices.
Toxicological evaluations and risk assessments for material safety and device biocompatibility.
Required for CE marking, FDA 510(k) submissions, and global market entry.

Edit Content
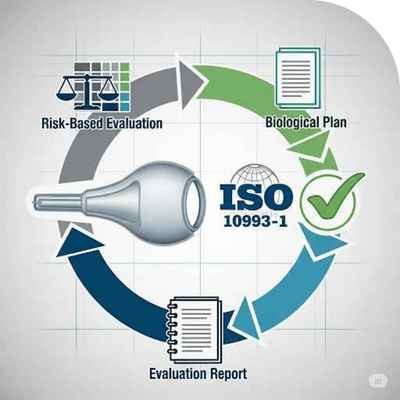
Risk-Based Evaluation, Biological Plan and Evaluation Report
We prepare ISO-compliant biological evaluation plans and reports for medical devices. Preparation of
Biological Evaluation Plans and Reports in accordance with ISO 10993-1. Required for biological
risk assessments of medical devices.
Edit Content
We determine regulatory identity and identify submission gaps across product categories.
Analysis to classify products (e.g., cosmetic vs. drug) and identify regulatory or data submission gaps.
Essential for correct regulatory strategy and compliance.

Edit Content
We standardize compliance with robust documentation tailored to your operations.
Creation of internal policies, standard operating procedures, and regulatory-ready dossiers. Required
for GMP, ISO, and regulatory audits.

Edit Content
We compile regulatory submissions with expert-developed CTD modules and summaries.
Development of Common Technical Document (CTD) modules for pharmaceuticals and APIs.
Required by FDA, EMA, PMDA, and other global authorities.

Edit Content
We minimize testing burden through expert toxicological justifications and waiver strategies
Expert-authored toxicological statements supporting testing waivers or alternative safety arguments.
Recognized under ICH, REACH, and ISO frameworks to minimize redundant testing.

10
+
Years Experience
395
+
Projects Done
50
+
Portfolios
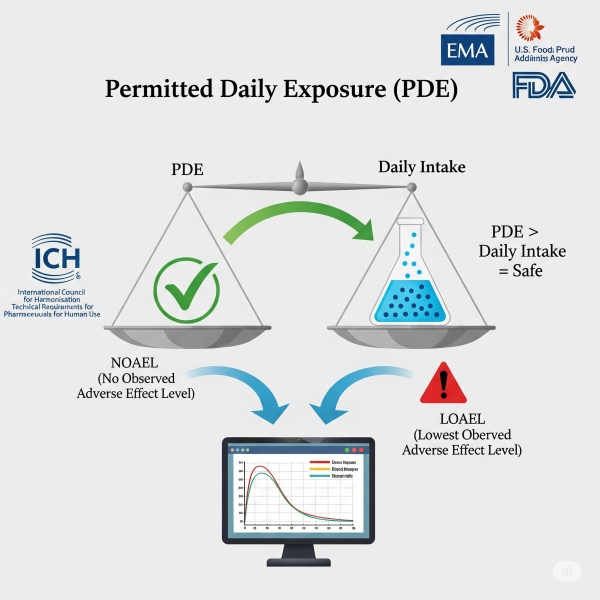
Permitted Daily Exposure (PDE) Determination
- Permitted Daily Exposure (PDE) is the maximum acceptable intake of a residual substance per day without health risk. It is a critical benchmark in pharmaceutical and chemical risk assessments.
- PDE limits are mandated by regulatory agencies like EMA, FDA, and ICH Q3C/Q3D. They ensure safety in drug manufacturing, preventing cross-contamination and toxic exposure.
- PDE is essential for calculating cleaning validation limits in pharmaceutical production. It ensures that residues do not pose a toxicological risk to patients.
- Establishing PDE is a core part of regulatory toxicology and safety dossiers. It supports global market access by complying with health authority expectations.
- VIVISCIENCES delivers expert PDE reports backed by robust toxicological data and regulatory insight. Our services help you meet compliance and accelerate approvals.
Plan to Start a Project


